| |
|
|
| |
Press release: PIC/S Meetings in Toyama (Japan)
From 11 to 15 November 2019, Japan’s Ministry of Health, Labour and Welfare (MHLW) and Pharmaceuticals and Medical Devices Agency (PMDA) hosted the following events in Toyama (Japan): PIC/S Committee meeting, PIC/S Executive Bureau meeting and PIC/S Annual Seminar.
|
| |
HEADLINES
|
- New PIC/S Chairperson, new Executive Bureau and new Sub-Committee Office Holders for 2020-21
- Revision of PIC Scheme
- Italy (Veterinary Agency) / DGSAF joins PIC/S
- PIC/S 2019 Annual Seminar on “Quality Assurance of Sterile Medicinal Products – Annex 1” hosted by Japan / MHLW & PMDA draws close to 160 inspectors from approximately 50 countries and areas
- PIC/S priorities and workplan for 2020
- New features for PIC/S Inspectorates’ Academy (PIA) and other updates in the field of PIC/S training activities and Expert Circles
- New Draft PIC/S Recommendation on How to Evaluate / Demonstrate the Effectiveness of a Pharmaceutical Quality System in relation to Risk-based Change Management and other PIC/S developments in the field of GM(D)P harmonisation
- Completion of PIC/S pre-accession of Russian Federation / Minpromtorg Russia, FSI SID&GP and Roszdravnadzor
- New PIC/S Pre-Accession Guidelines
- Bilateral meetings with China / NMPA and ICH
|
PIC/S COMMITTEE MEETING
|
|

|
|
The PIC/S Committee met on 11-12 November 2019, under the chairmanship of Mr Boon Meow Hoe (Singapore’s Health Sciences Authority / HSA). The meeting was attended by 40 out of 52 PIC/S Participating Authorities (PA) as well as by a number of Applicants, Pre-Applicants, and Associated Partner Organisations.
|
|
NEW PIC/S CHAIRPERSON
AND EXECUTIVE BUREAU
|
|
The PIC/S Committee elected Ms Anne Hayes (Ireland / HPRA) as Chairperson for the period 2020-2021. Ms Hayes has been an active member of the PIC/S Committee since 2005. She has also served as a PIC/S Executive Bureau member since 2013, in particular as Chair of the Sub-Committee on Compliance, which is one of the largest PIC/S Sub-Committees, in charge of compliance with the PIC Scheme.
|
|
 |
|
She will be the second PIC/S Chairperson from Ireland. The incoming Chairperson thanked the Committee and paid tribute to the outgoing Chairman, Mr Boon Meow Hoe (Singapore / HSA).
|
|
Outgoing PIC/S Chairman (2018-2019), Mr Boon Meow Hoe (Singapore / HSA), welcoming incoming PIC/S Chairperson (2020-2021), Ms Anne Hayes (Ireland / HPRA).
|
|
|
Mr Boon is the visionary of the PIC/S Inspectorates' Academy (PIA) and his chairmanship was marked by a significant expansion of PIC/S’ outreach and activities, in particular through efforts in the field of mutual reliance. PIC/S membership also increased by a new Member and relations with non-Members expressing interest in joining PIC/S were successfully strengthened during his term (2018-2019), notably through high-level discussions with China / NMPA and India / CDSCO.
The PIC/S Committee elected as incoming Deputy Chairperson for the period 2020-2021, Mr Paul Gustafson (Canada / ROEB), who will continue to chair the Sub-Committee on GM(D)P Harmonisation (SCH), in charge of all PIC/S activities in the field of harmonisation of GM(D)P.
The incoming Chairperson paid also tribute to the current Chair of the Sub-Committee on Communication (2016-2019), Mr Mark Birse (UK / MHRA), who will be leaving his Agency. Mr Birse actively contributed as Executive Bureau and Committee Member to PIC/S recent development, in particular with regard to the PIC/S Inspection Reliance Initiative.
|
A new PIC/S Executive Bureau was elected by the Committee as from 1 January 2020. The Executive Bureau Members for the period 2020- 2021 are: |
|
• Ms Anne Hayes (Ireland / HPRA), PIC/S Chairperson;
• Mr Paul Gustafson (Health Canada), PIC/S Deputy Chairman and Chair of the Sub-Committee on GM(D)P Harmonisation (SCH);
• Mr Boon Meow Hoe (Singapore / HSA), Immediate Past PIC/S Chairman;
• Mr Jacques Morénas (France / ANSM), Chair of the Sub-Committee on Training (SCT);
• Ms Susan Laska (US FDA), Chair of the Sub-Committee on Strategic Development (SCSD);
|
• Dr Andreas Krassnigg (Austria / AGES), Chair of the Sub-Committee on Expert Circles (SCEC);
• Mr Ger Jan van Ringen (Netherlands / IGJ), Chair of the Sub-Committee on Budget, Risk and Audit (SCB);
• Ms Stephanie Anctil (Health Canada), Chair of the Sub-Committee on Compliance (SCC);
• Mr David Churchward (United Kingdom / MHRA), Chair of the Sub-Committee on Communication (SC COM).
|
| |
|
|
|
NEW PIC/S SUB-COMMITTEE OFFICE HOLDERS
|
The PIC/S Committee elected the Members, Deputy Chairs and Chairs of the PIC/S Sub-Committee structure for the period 2020-2021. Office holders were elected for the following seven Sub-Committees: Training (SCT); Expert Circles (SCEC); Strategic Development (SCSD); Compliance (SCC); GM(D)P Harmonisation (SCH); Budget, Risk and Audit (SCB) and Communication (SC COM). All Sub-Committee Chairs will be Members of the PIC/S Executive Bureau. Out of the more than 80 newly appointed Sub-Committee office holders, all continents are represented.
|
|
REVISED PIC SCHEME
|
|
The Committee approved a revision of the PIC Scheme (PICS 1/95 (Rev 6)).
The Scheme gives PIC/S' legal basis as a non-profit association under Swiss law and governs its organisation and functioning. It is the PIC Scheme – which is different from the PIC Convention which consists in a mutual recognition Treaty – that provides the informal and flexible arrangement between PIC/S Participating Authorities which has made the success of PIC/S since it was established in 1995 as an extension to the PIC Convention of 1970.
The last revision of the PIC Scheme dated back to 2012. The purpose of this revision is:
- To clarify the definition of Member (“Participating Authority”) and Non-Member wishing to apply for membership (“Competent Authority”);
- To describe PIC/S’ legal status and explain what PIC/S is, why it has established; and
- To streamline the language.
|
ITALY (VETERINARY AGENCY) / DGSAF
JOINS PIC/S
|
|
The PIC/S Committee invited Italy’s Directorate General for Animal Health and Veterinary Medicinal Products (DGSAF) to join the Scheme as from 1 January 2020.
Italy / DGSAF will become PIC/S’ 53rd Participating Authority. DGSAF submitted a complete membership application in August 2016. The PIC/S assessment process was a partial assessment, facilitated through the recognition of the EMA Joint Audit Programme (JAP) audit conducted in October 2015, in line with the Letter of Agreement between PIC/S and EU/EEA Heads of Medicines Agencies (HMA). An on-site visit took place on 14-18 January 2019 to observe an inspection on biological products in order to complete the assessment.
This on-site visit facilitated the review of the implementation of corrective actions and successful finalisation of the assessment. The Audit team recommended to the Committee to accept the PIC/S membership application of Italy / DGSAF, subject to post-approval commitments regarding some PIC/S specific requirements.
Along with other PIC/S Participating Authorities competent for veterinary medicines, the joining of Italy (Vet) / DGSAF as a new PIC/S Member will further expand the outreach of PIC/S in the GMDP veterinary field.

|
The representative of Italy / DGSAF, Dr Virgilio Donini (3); with the PIC/S Chairman, Mr Boon Meow Hoe (Singapore / HSA) (2); the PIC/S Deputy Chairperson, Ms Anne Hayes (Ireland / HPRA) (5); the Rapporteur, Dr Jason Todd (UK / VMD) (1); and Co-Rapporteur, Mr Grégory Verdier (France / ANSES-ANMV) (4).
|
2019 PIC/S ANNUAL SEMINAR ON
QUALITY ASSURANCE OF STERILE MEDICINAL PRODUCTS – ANNEX 1
|
|

Councillor, Minister’s Secretariat,
Ministry of Health, Labour and
Welfare (MHLW), Mr Kazuhiko Mori
|
The 2019 PIC/S Annual Seminar was organised by Japan’s Ministry of Health, Labour and Welfare (MHLW) and Pharmaceuticals and Medical Devices Agency (PMDA) in Toyama (Japan) on 13-15 November 2019. The topic of the Seminar was “Quality Assurance of Sterile Medicinal Products – Annex 1”. Seminar participants were invited by Councillor, Minister’s Secretariat, Ministry of Health, Labour and Welfare, Mr Kazuhiko Mori.
|
|

Governor of the Toyama Prefecture, Mr Takakazu Ishii
|
The Seminar was opened by the Governor of the Toyama Prefecture, Mr Takakazu Ishii, along with the Senior Executive Director of PMDA, Dr Yoshikazu Hayashi and the PIC/S Chairman, Mr Boon Meow Hoe. The Seminar, which was the first organised in Japan since MHLW, PMDA and the Japanese Prefectures joined PIC/S in 2014, was attended by close to 160 inspectors from approximately 50 countries and areas. All continents were represented.
|

Senior Executive Director of
Pharmaceuticals and Medical
Devices Agency (PMDA),
Dr Yoshikazu Hayashi |
Annex 1 (manufacture of sterile medicinal products) of the PIC/S GMP Guide is currently under revision. The revision is led by a PIC/S-EMA Working Group with WHO on the revision of Annex 1 chaired by France / ANSM. It is a key revision to the PIC/S and EU GMP Guides and following a focused stakeholder consultation in 2018 the Working Group successfully reviewed more than 6,300 comments.
|
|
|
The Seminar, through a mix of presentations and workshops, provided participants with a further understanding of Annex 1, based on issues discussed during its ongoing revision and through case studies of sterility assurance. In addition to the revision of Annex 1, the programme covered topics related to Process Simulation Test (PST), Clean Room Management and Clean Room Qualification and Environmental Monitoring, Management of Water for Injection (WFI), Quality Control and Stable Supply of Single-Use Systems, Restricted Access Barriers Technology (RABS) and Isolators as well as Rapid Microbiological Methods.
The Seminar also allowed participants to acquire skills on how to make risk based decisions during GMP inspections of sterile products and discuss examples of observations for sterile products. It was an ideal opportunity for both novice and experienced inspectors to enhance their inspection skills through knowledge sharing and discussion. For the very first time in a PIC/S Seminar, active discussions were stimulated through the innovative use of videos taken of practical manufacturing operations, screened and discussed during the Seminar workshops.
The Seminar outcome led to the need for PIC/S to establish in the future a new Expert Circle dedicated to Sterile Manufacturing as well as a new Working Group on Emerging New Technologies.

For more information on the Seminar, see below.
|
PIC/S PRIORITIES AND WORKPLAN FOR 2020
|
|
The Committee reviewed and discussed the work plans of PIC/S 7 Sub-Committees which cover all areas of PIC/S activities: organisational, compliance, training and expert discussions, harmonisation of GM(D)P, strategic development and co-operation, communication and financing.
These work plans – which are aligned with the goals and priorities as defined in the PIC/S Road Map for the period 2018-20 – have been consolidated into a PIC/S 2020 Work Plan which is published on the page “Publications”.
|
Interview of the new PIC/S Chairperson (2020-2021)
Ms Anne Hayes (Ireland / HPRA)
 |
|
Q. What are PIC/S key objectives and priorities for 2020?
A. The primary focus for early 2020 will be the outcome of the launch of the PIC/S Inspection Reliance Initiative. This initiative aims at strengthening mutual reliance between Competent Authorities. Its purpose is to encourage reliance based on desk-top assessments of existing inspection reports rather than duplicate foreign GMP inspections. PIC/S Participating Authorities have been invited to collect statistics during 2019 on such desk-top assessments in connection with the implementation of the PIC/S Guidance on GMP Inspection Reliance (PI 048-1).
|
|
|
Means to progressively increase the use and operation of this reliance framework will be discussed in 2020. A pilot on the sharing of information on borderline cases will be undertaken. PIC/S will also further engage with Industry on this Initiative, in particular following interest expressed by several stakeholders, such as GPMLF. The active support of Industry is important as it is up to Industry to proactively share reports to avoid duplicate inspections.
Another key priority for 2020 is building up the PIC/S Inspectorates’ Academy (PIA) and ensuring active involvement of all competent PIC/S bodies. This is a flagship project of PIC/S, its potential is very significant also to mutual trust and reliance. Harmonisation and calibration of training and qualification of GMP Inspectors are essential to ensuring high-quality and comparable GMP inspections.
In 2020, PIC/S will see its membership increase to 53 PIC/S PA with the Italian Veterinary Agency DGSAF acceding to the Scheme. PIC/S also aims to strength co-operation and engagement in the field of GMP with its current Partners (i.e. the EMA, EDQM, UNICEF and WHO) and other relevant Organisations (ASEAN, ICH, European Commission, etc.). PIC/S will welcome a new Partner in 2020: the World Organisation for Animal Health (OIE). This new co-operation will strengthen PIC/S involvement in the field of VMPs. Other fields in which PIC/S will continue to strengthen its involvement are e.g. GDP, ATMP and IMPs. I trust PIC/S activities will continue to expand in 2020 through the remarkable work carried out by its relevant Expert Circle and Working Groups and that we will continue to deliver on high quality GMDP guidance in line with PIC/S’ mission to lead the international development, implementation and maintenance of harmonised GMP standards and quality systems of inspectorates in the field of medicinal products.
Q. How do you see PIC/S evolving in the future?
A. PIC/S has evolved tremendously these last few years, in particular since its last reorganisation in 2014, which introduced the Sub-Committee structure. In addition to the PIC/S Committee, Executive Bureau and Secretariat, PIC/S comprises 7 Sub-Committees (SC), regrouping more than 80 SC members; 5 Expert Circles regrouping more than 25 Co-ordinating Committee members; 15 Working Groups (WG) regrouping more than 100 members; and 2,000 inspectors to which it aims at offering training via PIA. The expansion of PIC/S membership, scope of activities, output and outreach – which is set to continue - have led to the need to further strengthen the Organisation. This is one of the main goals highlighted in the Road Map (2018-20) – besides training inspectors and sharing information. As PIC/S is now part of a wider international network of organisations and is a leader in the global landscape of ensuring pharmaceutical quality through inspection and compliance, it must adapt to meet demands and future challenges to support its Members and secure PIC/S’ role internationally. In this perspective, PIC/S will continue to improve communication – which will also include developing tools to measure utilisation and implementation of PIC/S guidance documents - identify and implement activities to raise the profile and prominence of PIC/S at a global level; enhance PIC/S’ SC structure; strengthen the PIC/S Secretariat and identify new income streams, which will yield the required funding necessary to finance PIC/S’ projects.
Q. What are the upcoming milestones for PIC/S?
A. PIC/S will be celebrating its 50th anniversary in May 2021. This milestone will be marked by a special anniversary event combined with the PIC/S 2021 Seminar, which will be hosted by Ireland / HPRA in Dublin (Ireland) on 31 May – 4 June 2021. This will be an occasion to reflect on PIC/S accomplishments over the past 50 years, discuss contemporary issues and PIC/S future, outlining the future guiding lines of a new Road-Map for PIC/S as from 2021.
Other milestones include the accession of future Members, once they have reached a GMP regulatory control and Quality System equivalent to PIC/S. The membership expansion of PIC/S is set to further grow in the next few years, in particular with regard to some global players in the field of pharmaceutical inspections, such as Brazil which is close to the completion of its PIC/S accession process, Russia which has just completed its PIC/S pre-accession process; and China and India with which PIC/S has engaged in discussions and actively encourages to apply for pre-accession or accession.
|
NEW FEATURES FOR
PIC/S INSPECTORATES’ ACADEMY
|
|
The PIC/S Inspectorates’ Academy (PIA) is a global capacity building and training initiative developed by PIC/S Participating Authorities aiming at delivering inspection excellence through harmonised training in the field of Good Manufacturing Practices (GMP) to PIC/S currently approx. 2,000 inspectors.
Harmonisation and calibration of training and qualification of GMP Inspectors is key to having strongly aligned and comparable competencies and capacities across National Competent Authorities (NCA)’ inspectorates on quality manufacturing and GMPs worldwide. This is key to mutual trust which in turn will allow for mutual reliance between NCA in line with PIC/S Guidance on GMP Inspection Reliance (PI 048-1). This will allow not only to maximize inspectional resources but also to strengthen the protection of public health by ensuring effective, high-quality and comparable GMP inspections for the quality of regulated pharmaceutical products.
The ultimate goal of PIA is to harmonise and standardise GMP training and qualification process at an international level through a recognised system matching a high grade of skills. Such a system should ensure that inspectors apply GMP consistently so that high quality standards for medicinal products are met worldwide in the interest of public health. This will also help pool together training resources for PIC/S PA and thus contribute to cost savings for Agencies while strengthening co-operation, harmonisation and resource sharing in the field of training.
At its meeting in Toyama, the Committee endorsed in principle several documents drafted by the PIC/S Sub-Committee on Training (SCT), led by Jacques Morénas (France / ANSM), which will be key in the future development of PIA and its features. These documents outline an architecture for a harmonised PIA Training Programme (including its related training Curricula and Cycle) and Qualification Process.
The PIA Training Programme aims to define harmonised minimum training requirements and its related curricula to define the scope of training for concrete inspector’s competence in specific fields (e.g. API, sterile, biologicals, etc.). The architecture clarifies the roles and delimits the responsibilities between the PIA, PIC/S Participating Authorities (including Tutors) and Trainees. Training will be based on high quality training materials focused on GMP requirements and inspection skills, to be delivered through various formats. The formats are to be provided either through training tools offered by PIA or by the PIC/S Participating Authorities or both.
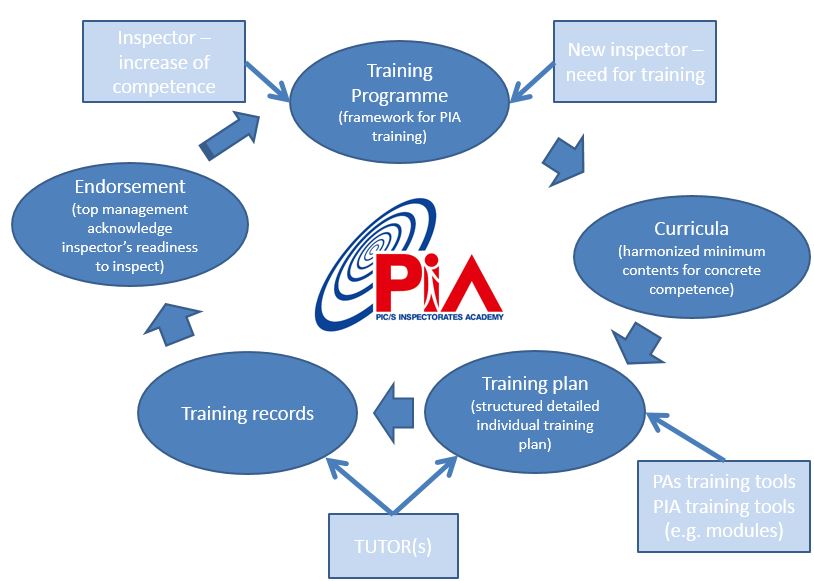
Outline of PIA Training Cycle
The Committee also endorsed in principle a first Draft Training Curriculum on Quality Risk Management (QRM) developed by the PIC/S Expert Circle on QRM and welcomed a pilot demo of an e-learning module on QRM developed on this basis. This pilot will serve as model for the development of future e-learning modules part of the PIA Training Programme, which will be operated by a Learning Management System (LMS) to be established in co-ordination with interested PIC/S Participating Authorities.
For more information on PIA, see general presentation brochure on PIA.
|
OTHER DEVELOPMENTS IN THE FIELD OF TRAINING
AND EXPERT CIRCLES
|
|

|
|
The Committee was updated on the programme of the 2020 PIC/S Annual Seminar on "How to be a Good GMP Inspector in 2020" which will be hosted by Thailand’s Food and Drug Administration (Thai FDA) in Bangkok (Thailand) from 18-20 November 2020. To view the promotional video click here.
|
|
|
The Committee also discussed preparations for the 2021 PIC/S Annual Seminar which will be hosted by Ireland / HPRA in Dublin (Ireland) in May 2021 and will be combined with PIC/S 50th Anniversary (see interview above of the new PIC/S Chairperson 2020-21).
The 2022 PIC/S Annual Seminar will be hosted by Finland / FIMEA and the 2023 PIC/S Annual Seminar by Korea (Republic of) / MFDS.
The Committee welcomed a report on the operation of the PIC/S Joint Visits Programme (JVP) for conducting Good Clinical Practice (GCP) and Good Pharmacovigilance Practice (GVP) inspections, which is co-ordinated by the PIC/S GCP and GVP Working Group, led by UK / MHRA.
The report outlined the positive feedback received about the JVP, indicating that it provides a valuable learning opportunity and an effective way of sharing experience and best practice. Some Inspectorates have also provided feedback that what they have learned during Joint Visits has been beneficial in the development of their inspection programmes.
|
|
The PIC/S Joint Visits Programme and its expansion into the field of GCP and GVP
Under the PIC/S Joint Visits Programme, three Inspectors from three different countries are teamed up to observe GMP inspections in each country with a view to comparing inspection procedures and techniques and to harmonise GMDP interpretation.
In the field of GCP and GVP, the purpose of the JVP programme is to:
- provide further training for GCP and GVP inspectors through the exchange of experience between them;
- provide the means of harmonising inspection procedures and developing inspection guidance; and
- ensure and maintain mutual confidence between inspectors of PIC/S Participating Authorities as well as with GCP and GVP inspectors of non-PIC/S Participating Authorities.
Differences and similarities in inspection procedures and techniques are reported to the PIC/S Working Group after each inspection in the form of a short report of the Joint Visit.
|
|
The Committee discussed the status of a survey on future training priorities for PIC/S, taking also into account priorities identified by EMA / HMA (EEA Heads of Medicines Agencies). The integration of these priorities in future training activities and in particular the PIC/S Inspectorates’ Academy (PIA) will be carried out through closer co-operation between the PIC/S Sub-Committee on Training (SCT) and the PIC/S Sub-Committee on Expert Circles (SCEC). This will ensure training priorities are fed into the training activities of PIC/S Expert Circles.
Expert Circles and relevant Working Groups will also become more actively involved in the development and design of training curricula, training materials and their continuous updating under PIA in their respective fields of competence, including how to best share knowledge and deliver ongoing training to inspectors who cannot attend face-to-face Expert Circle training events.
|
Recent PIC/S Expert Circle meetings and training activities include:
|
|

|
|
The 1st PIC/S Expert Circle on Cross-Contamination in Shared Facilities (CCCISF) was held on 19-21 June 2019 in Taipei (Chinese Taipei). The meeting was designed to share and develop understanding of challenges facing inspectors on inspection of risk of (chemical) cross contamination in shared facilities and the inspection approaches required to successfully inspect for compliance with PIC/S GMP. 82 participants attended from 20 countries.
|
|
|
The 9th PIC/S Expert Circle meeting on APIs was held in Madrid (Spain), on 7-9 October 2019, hosted by Spain / AEMPS. The focus of this meeting was to provide advanced training to GMP inspectors in the field of API inspections, in topics related to the commercialization of APIs, emerging technologies and other current matters such as the revision of the PIC/S Aide-Memoire on API Inspections. 67 participants from 38 countries took part.
|
|

|
|
|

|
|
The 25th PIC/S Expert Circle meeting on Human Blood, Tissues, Cells and ATMPs was held in Jakarta (Indonesia), on 8-10 October 2019, hosted by Indonesia / NADFC. The focus of this meeting was "Blood, Tissues, Cells and ATMPs: How to Inspect and Current Trends" and consisted in presentations and workshops. The meeting was attended by 94 participants from 27 authorities.
|
|
|
For future PIC/S training events, see below.
|
NEW DRAFT PIC/S RECOMMENDATION ON
HOW TO EVALUATE / DEMONSTRATE THE EFFECTIVENESS OF A PHARMACEUTICAL QUALITY SYSTEM IN RELATION TO RISK-BASED CHANGE MANAGEMENT
|
|
The PIC/S Expert Circle on Quality Risk Management (QRM) has developed a new draft Recommendation on How to Evaluate / Demonstrate the Effectiveness of a Pharmaceutical Quality System in relation to Risk-based Change Management (PI 054-1 (Draft 1)).
This draft Recommendation will be applied on a 6-month trial basis by PIC/S Participating Authorities.
The purpose of this draft document is to provide guidance on evaluating and demonstrating the effectiveness of a PQS in relation to risk-based change management. This is in recognition of the fact that the PIC/S GMP Guide requires companies to demonstrate the effectiveness of their PQS and to apply quality risk management (QRM) principles to change control activities. This document is very important considering the future implementation of ICH Q12. Further information on the background to this Recommendation and the anticipated benefits of this guidance are provided in PIC/S Concept Note (PS/INF 88/2019).
This draft document is not open for comments by industry. Formal adoption of the guidance will proceed in accordance with PIC/S procedures on a revised version after completion of the trial-period.
The draft Recommendation (PI 054-1 (Draft 1)) and the Concept Note (PS/INF 88/2019), are published under “Publications”.
|
Interview of the Chair of the PIC/S Exert Circle on Quality Risk Management (QRM), Dr. Kevin O’Donnell (Ireland / HPRA)
|

|
Q. How did the idea of this new PIC/S guidance come about and in response to what needs has it been developed?
A. This topic has not been the subject of much guidance in the past, and working on it was considered a natural progression of the PIC/S Expert Circle’s existing QRM training activities, which for several years have included risk-based change management as a topic. The effectiveness of the Pharmaceutical Quality System (PQS) is an important element of the PIC/S GMP requirements, but until now there has not been much practical guidance on how to assess that.
|
|
|
In addition, given the recent finalisation of the ICH Q12 guideline, which positions PQS effectiveness in relation to risk-based change management as a core concept, there was an opportunity within the Expert Circle on QRM to develop guidance that could help with the implementation of Q12. Work began during a PIC/S QRM Expert Circle meeting in Taipei in September 2018, during which the input of industry representatives was taken into account and contributed to the development of this document.
Q. What is the scope of this draft guidance and its key points?
A. The document applies to GMP inspections of manufacturers of medicinal products and active pharmaceutical ingredients. It provides practical guidance for GMP inspectors when seeking to evaluate the effectiveness of a company’s PQS in relation to risk-based change management. It is important to note that the document does not suggest or create any new GMP expectations or requirements. The key points in the document cover all relevant steps in the change management process – from change proposal, change assessment, change planning & implementation, through to change review and effectiveness checks, and they indicates within each step the aspects that render the PQS to be effective in that area.
Q. This new draft guidance foresees several anticipated benefits - what are these?
A. I think that the document will deliver benefits in several areas. For example, it will hopefully help GMP inspectors better understand what risk-based change management means at a practical level, and how it might be evaluated during inspection. This is of practical relevance to Inspectors, because the PIC/S GMP Guide requires companies to demonstrate the effectiveness of their PQS, and change management is expected to be a major component of the PQS. In addition, the PIC/S GMP Guide requires companies to use risk-based approaches to the management of changes. Therefore, PQS effectiveness and risk-based change management go hand-in-hand and indeed, they complement each other.
There are other benefits envisaged too. ICH Q10 sets out the potential for risk-based regulatory oversight for companies which demonstrate that an effective PQS is in place. ICH Q10 indicates that, if the principles and concepts of ICH Q8, Q9 & Q10 are adopted, and if the effectiveness of the PQS is demonstrated, then there may be new opportunities for risk-based regulatory oversight and flexibility in reporting requirements that will promote lifecycle continual improvements. This new guidance provides very practical guidance which companies can use to proactively demonstrate the effectiveness of their PQS in an area that is of relevance to all pharmaceutical manufacturers – risk-based change management. The Expert Circle considers that the application by a company of the guidance in this document will provide evidence of the effectiveness of the PQS at that company in relation to risk-based change management. This is important not only in the context of GMP, it should also help companies achieve or realise the opportunities presented by ICH Q10 and Q12.
There is also an indirect benefit for patients - if such a risk-based change management system were in place within a company’s PQS, it should lead to the timely management of risks to patient safety, as well as better quality and manufacturing performance, continual improvement and innovation.
Q. What are the next steps in the development and adoption process for this draft guidance?
A. At the end of the 6-month application on a trial-basis, comments received by PIC/S PA will be reviewed and the document updated as needed prior to it being submitted for adoption by PIC/S. This document will also help form the basis for a revision of the current PIC/S Aide Memoire on QRM relating to inspection of quality risk management activities.
|
|
LATEST PIC/S DEVELOPMENTS IN THE FIELD
OF GM(D)P HARMONISATION
|
The PIC/S Committee was updated on:
- the revision of Annex 1 (manufacture of sterile medicinal products) of the PIC/S and EU GMP Guide, by the PIC/S-EMA Working Group with WHO on Annex 1, led by France / ANSM, and on planning by the PIC/S Sub-Committee on GMPD Harmonisation (SCH) of revisions to other PIC/S guidance documents which will be impacted by the revision of Annex 1;
- the status of the revision of Annex 2 (manufacture of biological medicinal substances and products for human use) of the PIC/S GMP Guide. This revision developed by the PIC/S Working Group established with WHO, led by Australia / TGA, was subject to a focused stakeholder consultation which included both consultation questions and an opportunity to comment on:
- a draft Annex 2A (PS/INF 25/2019 (Rev. 1)): Manufacture of Advanced Therapy Medicinal Products for Human Use; and
- a draft Annex 2B (PS/INF 26/2019 (Rev. 1)): Manufacture of Biological Medicinal Substances and Products for Human Use.
Draft Annex 2A takes into account the international development in the regulation of Advanced Therapy Medicinal Products (ATMP) with particular attention to the European Commission guideline on GMP for ATMP which has been published since the latest revision of the EU Annex 2, while addressing at the same time concerns of PIC/S Participating Authorities with regard to patient safety and proportionate regulation for ATMPs. Draft Annex 2B is the revised version of EU Annex 2 for biologics (excluding ATMPs). For more information on the stakeholder consultation which ended on 20 December 2019 see News.
- step 2 of the PIC/S adoption process (Consultation of non-EEA PA of their national industry association(s)) on the revision of Annex 13 (manufacture of investigational medicinal products), which ended in November 2019.
- progress in the revision of the PIC/S guidance documents for blood prepared by the PIC/S Working Group, led by Switzerland / Swissmedic, namely:
- the draft revision of PIC/S Good Practice Guidelines for Blood Establishments and Hospital Blood Banks (PE 005-4). The draft revision is based on the Good Practice Guidelines (GPG) for blood establishments drafted by the European Directorate for the Quality of Medicines & HealthCare of the Council of Europe (EDQM) and the European Commission. Step 1 (PIC/S-internal consultation) ended in April 2019. The Committee supported advancing to step 2 (Consultation of non-EEA PA of their national industry association(s));
- the draft revision of the PIC/S Guide to Inspections of Source Plasma Establishment and Plasma Warehouses (PI 008-4) which has undergone a complete revision in order to align it with the revision of PE 005-4. It has been converted into an Aide-Memoire which will be advanced for step 1 (PIC/S-internal consultation).
- progress in the review by the Working Group on Data Integrity, co-led by UK / MHRA and Australia / TGA, on the outcome of the focused stakeholders’ consultation on the revised draft PIC/S guidance on Good Practices for Data Management and Integrity in Regulated GMP/GDP Environments (PI 041-1 (Draft 3)), which ended on 28 February 2019;
- planning of Step 3 (adoption) of a PIC/S Aide Memoire on Inspection of Data Management and Integrity and of a PIC/S Aide Memoire on PIC/S Data Integrity System-Specific Guidance, which should be published once adopted by the Committee, in line with the need for their access to be restricted to PIC/S Members-only;
- outcome of Step 1 (internal consultation of PIC/S PA) of the revision of the PIC/S SOP on Inspection Report Format (PI 013-4) in order to better align with the Guidance on Classification of Deficiencies (PI 040-1) which entered into force on 1 January 2019;
- progress in the revision of the PIC/S Guide to good Practices for the preparation of medicinal products in Healthcare Establishments (PE 010-4) including, as previously endorsed by the Committee, the EMA / EU Parenteral Nutrition guidance as an appendix;
- outcome of Step 1 (internal consultation of PIC/S PA) of the PIC/S Aide-Memoire on Inspection of Health Based Exposure Limit (HBEL) Assessments and use in Quality Risk Management (PI 052-1) and the Questions and Answers on Implementation of Risk-based Prevention of Cross-contamination in Production and Guideline on Setting Health-Based Exposure Limits for Use in Risk Identification in the Manufacture of Different Medicinal Products in Shared Facilities (PI 053-1), developed by the Expert Circle on Controlling Cross-Contamination in Shared Facilities (CCCISF), led by UK / MHRA.
|
The PIC/S Committee discussed:
|
The PIC/S Committee endorsed:
- a recommendation by the SCH to not transpose for PIC/S purposes the new EU Annex 21 (Importation of medicinal products), for which PIC/S is represented in the EMA Drafting Group.
|
The PIC/S Committee adopted:
- a new mandate for the Working Group on the revision of the PIC/S guidance documents for blood;
- a new mandate for a new Working Group on Controlling Cross-Contamination in Shared Facilities (CCCISF), which will operate along with the Expert Circle on CCCISF.
|
The PIC/S Committee noted:
- PIC/S discussions with ICH Management Committee as well as involvement in ICH Expert Working Groups, in particular Q13, M10 and E19;
- the status of the Working Group for a PIC/S Aide Memoire on Tissues and Cellular Therapy Products Inspections;
- the ongoing status of development of:
- a draft PIC/S Aide-Memoire for GDP Inspections and Q&A on GDP;
- a revision of PIC/S Recommendations on Validation Master Plan; Installation and Operational Qualification; Non-sterile Process Validation and Cleaning Validation (PI 006-3);
- a revision of PIC/S Aide-Memoire on Quality Control Laboratories (PI 023-2);
- a revision of PIC/S Aide-Memoire on the Inspection of APIs (PI 030-1).
|
COMPLETION OF PRE-ACCESSION PROCESS
FROM THE RUSSIAN FEDERATION
|
|
The Russian Federation / Minpromtorg Russia, FSI "SID&GP" and Roszdravnadzor (under the Russian Ministry of Health) completed their PIC/S pre-accession procedure lodged on 28 August 2017. The Committee endorsed a recommendation by the Rapporteur that the Russian Federation / Minpromtorg and FSI "SID&GP" as well as Roszdravnadzor are now ready to apply for PIC/S membership.

|
|
|
NEW PIC/S PRE-ACCESSION GUIDELINES
|
|
The Committee adopted new PIC/S Pre-Accession Guidelines developed by the Working Group on drafting Pre-Accession Guidelines, led by France / ANSM.
These new Guidelines ensure a new approach for the pre-accession process in which a gap-analysis is no longer performed. This new approach is based on the experience that past pre-accession processes have proven to be time- and work consuming, taking as long as a paper assessment under the accession process. Rather than duplicating tasks (assessment under pre-accession and assessment under accession), PIC/S decided to change the purpose of the pre-accession process, to consist in explaining in detail to Pre-Applicants what PIC/S requirements exactly entail, in particular those listed in the PIC/S Audit Checklist.
This new pre-accession procedure therefore serves to identify the necessary elements part of the GMP Regulatory Compliance Programme as defined in the Audit Checklist to be provided by the interested National Competent Authority to help it understand the gaps between PIC/S requirements and its GMP Regulatory Compliance Programme. It is left up to the Pre-Applicant to assess the gap, based on his/her understanding of the PIC/S requirements as explained during the process.
The new Pre-Accession Guidelines are published under Publications.
|
PIC/S MEMBERSHIP APPLICATIONS
|
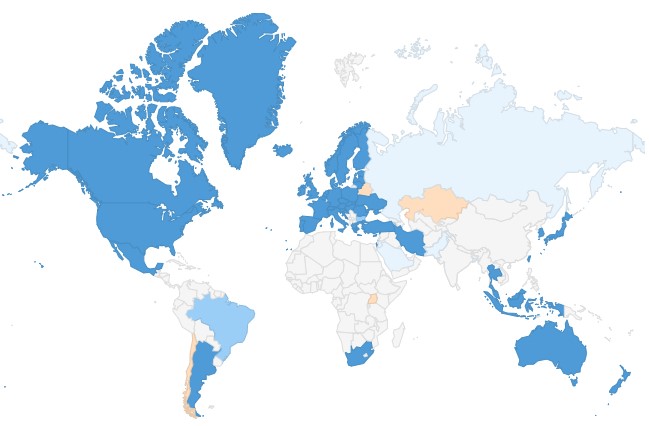 |
Overview of PIC/S 53 Members as of 1 January 2020 (dark blue); 3 Applicants (medium blue); 5 (Pre-) Applicants (pale blue) and 4 Former Pre-Applicants (pale orange)
|
| |
Applicants (3)
|
Pre-Applicants (5)
|
| |
Armenia / SCDMTE
Brazil / ANVISA
Bulgaria / BDA
|
Bangladesh / DGDA
Jordan / JFDA
Pakistan / DRAP
Russian Federation / Minpromtorg
Russia, FSI "SID&GP" and Roszdravnadzor*
Saudi Arabia / SFDA*
|
|
|
* presently former Pre-Applicants
|
|
- An update was given on the status of assessment of the membership application of Armenia / SCDMTE;
- The Committee was updated on the on-site visit for the membership application of Brazil / ANVISA, which took place on 9-21 October 2019. The assessment report will be provided at the next Committee meeting;
- The planning of an on-site visit combined with a Canadian MRA assessment was agreed for Q1 of 2020 for the membership application of Bulgaria / BDA which will be facilitated through a partial assessment taking into account the audit performed within the EMA Joint Audit Programme, in line with the PIC/S-EEA HMA Letter of Agreement;
- An update was given on the status of the assessment of the pre-accession application of Bangladesh / DGDA, which is ongoing;
- An update was also given on progress in the assessment of the pre-accession application of Jordan / JFDA. The pre-accession gap-analysis report will be provided at the next Committee meeting;
- The Committee was updated on the status and progress in the assessment of the pre-accession membership application of Pakistan / DRAP, which was represented at the meeting by its new Head of Agency;
- The Committee was updated by Saudi Arabia / SFDA on their plans to imminently lodge a membership application further to the completion of the pre-accession process, which was closed on 10 April 2019.
|
BILATERAL MEETINGS WITH
CHINA / NMPA AND ICH
|
|
A bilateral meeting between the PIC/S Executive Bureau and a high-level Delegation from China's National Medical Products Administration / NMPA (formerly CFDA) took place on 14 November 2019. This meeting allowed to make good progress in addressing some issues raised by NMPA in connection with a possible future PIC/S application or pre-application.
A bilateral meeting between the PIC/S Executive Bureau and the Chair of the Management Committee of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH), Dr Theresa Mullin (US FDA), took place on 13 November 2019. This meeting allowed to discuss the advantages and explore opportunities for mutually beneficial co-operation between both Organisations, notably in the field of training.
|
OTHER NEWS
|
The Committee:
- was updated on the outcome of discussions within the PIC/S Executive Bureau meeting on the morning of 11 November 2019, which preceded the Committee meeting;
- discussed the scope of a checklist to determine the use and implementation of PIC/S guidance documents;
- discussed and reviewed several new communication tools, including a stakeholder mapping tool;
- adopted a revised draft mandate for the PIC/S Working Group on Third-Party Funding and approved PIC/S’ 2020 Budget and upcoming 3-year Budget Plan;
- was updated on progress made by the PIC/S Working Group on Unique Facility Identifiers (UFI), led by US FDA; the PIC/S Working Group on Inspector Travel Safety, led by UK / MHRA; as well as the PIC/S Working Group on Confidential Informants, which is co-led by US FDA and UK / MHRA;
- discussed PIC/S Inspection Reliance Initiative and possible engagement with industry (see above interview of the PIC/S Chairperson 2020-21 on workplan and priorities for 2020);
- was updated on recent GMP-related developments by EMA, UNICEF and WHO as PIC/S Associated Partner Organisations as well as was updated on current developments with regard to the implementation of the EU - US Mutual Recognition Agreement (MRA);
- was updated by the PIC/S - ASEAN Liaison Authority, Singapore / HSA, on recent activities within ASEAN. The PIC/S Secretariat also reported to the Committee that the non-binding exchange of letters between PIC/S and the ASEAN Pharmaceutical Product Working Group (PPWG), which aims to allow for co-operation between PIC/S and the ASEAN PPWG in GMP matters, is in the signing process. The Committee discussed and endorsed a recommendation by the PIC/S Sub-Committee on Strategic Development (SCSD) that a non-binding exchange of letters should also be signed between PIC/S and the ASEAN Traditional Medicines Health Supplements Product Working Group (TMHS PWG), in order to establish a working relationship.
- noted that the informal exchange of letters as a basis for future co-operation with the World Organisation for Animal Health (OIE) was in the signing process;
- was given a presentation on the point of view of France / ANSM’s Inspection Division on ICH guideline Q12 (technical and regulatory considerations for pharmaceutical product lifecycle management);
- noted feedback from the PIC/S representative to ICH and that a presentation on PIC/S is to be given at the upcoming meeting of the International Pharmaceutical Regulators Programme (IPRP) in Singapore in November 2019;
- noted the status of the PIC/S Working Group on Quality Defects Procedures, in charge of transposing for PIC/S purposes the revised EMA procedures on (i) Managing Reports of Suspected Quality Defects in Medicinal Products; and (ii) Handling Rapid Alerts Arising from Quality Defects;
- noted progress made by the PIC/S Working Group on Veterinary Medicinal Products (VMP), led by France / ANSES-ANMV;
- was updated on completion of the draft Guideline and Interpretation of the Audit Checklist by the PIC/S Sub-Committee on Compliance, in co-operation with the EU Joint Audit Programme Compliance Group. This draft is under PIC/S and EMA internal consultation;
- was updated on completion of a draft Revision of PIC/S Accession Guidelines and related Documents by the dedicated Working Group, led by France / ANSM, which will be issued for PIC/S internal consultation;
- endorsed the report, subject to some minor amendments, for the PIC/S re-assessment of Switzerland / Swissmedic, which was combined with a MRA re-assessment by Health Canada and co-ordinated with the EMA JAP audit of Liechtenstein / AG, for which the on-site visit took place on 15-19 October 2018;
- was updated on the outcome of the PIC/S re-assessment of Canada / ROEB, for which the on-site assessment visit took place on 7-11 October 2019;
- discussed the situation of South Africa / SAHPRA and the planning of the PIC/S re-assessment, postponed to 2020;
- took note of a call for the Re-Assessment Teams for the PIC/S re-assessments of Indonesia / NADFC and New Zealand / Medsafe which will be taking place in 2020;
- was updated on corrective actions or updates by recently acceded PIC/S PA or PIC/S PA under Re-assessment;
- noted that the Working Group on the revision of PI 011-3 (Computerised Systems) is still on hold while awaiting the revision of Annex 11 (Computerised Systems) of the PIC/S – EU GMP Guide;
- noted the outcome of a side-meeting between the PIC/S Sub-Committee on Training (SCT) Chairman and WHO which took place in Geneva on 9 April 2019, in order to discuss opportunities for co-operation in the field of training between PIC/S and WHO;
- noted that the Committee is to reconvene in Geneva (Switzerland) on 21-22 April 2020.
|
COMING UP...
|
|

|
|
- 26-28 May 2020: PIC/S Expert Circle on Good Distribution Practice (GDP), in Kyiv (Ukraine), hosted by Ukraine / SMDC;
- 19-23 October 2020: 2020 Japan / PMDA - ATC GMP Inspection Seminar, with the support of PIC/S, hosted by Japan / PMDA (location to be confirmed);
|
| |
|
|
|
|

|
|
- 18-20 November 2020: Annual PIC/S Seminar on “How to be a Good GMP Inspector in 2020”, in Bangkok, hosted by Thailand / Thai FDA;
- dates to be confirmed in 2020: PIC/S New Inspector Training Course, Dublin (Ireland), hosted by Ireland / HPRA.
|
| |
|
|
|
2019 PIC/S ANNUAL SEMINAR ON
QUALITY ASSURANCE OF STERILE MEDICINAL PRODUCTS – ANNEX 1
|
|

|
|
Seminar Welcome Remarks by the Governor of Toyama Prefecture, Mr Takakazu Ishii
Mr Ishii welcomed all Seminar participants to Toyama, Japan’s city of medicine. He highlighted the development and expansion of Toyama’s pharmaceutical industry, which has a long history and tradition of over 300 years. The city is presently renowned for its advanced technologies.
|
|
Considering the importance of the pharmaceutical industry to Toyama, he underlined what it meant to be a host for the PIC/S Committee Meeting and the PIC/S Seminar held for the first time in Japan. He looked forward to PIC/S events giving further momentum to growth of the pharmaceutical industry in Toyama and wished all participants a successful Seminar with a view to contributing to improvement of GMP inspection technology and global health advancement.
|
|

|
|
Official Dinner Opening Remarks by the Chief Executive of PMDA, Dr Yasuhiro Fujiwara
Dr Fujiwara said it was a great honor to invite nearly 160 experts from the world to Japan and welcomed all participants on behalf of PMDA. He recalled that in July 2014, inspection authorities in Japan composed of MHLW, PMDA and 47 prefectural governments became a PIC/S Participating Authority. The following year
|
|
|
in June 2015 the “PMDA International Strategic Plan 2015” was established and publicized the plan of PMDA’s international activities.
Dr Fujiwara stated that as a result, our international activities in cooperation with PIC/S are steadily making outstanding outcomes now. Especially the high quality information shared among PIC/S member authorities has provided GMP inspection authorities in Japan with great advantages. Currently, PIC/S is regularly holding seminars and expert circles to share the latest scientific knowledge, and it enables us to share various information on quality of pharmaceuticals both rapidly and accurately. Although, PIC/S is a voluntary organisation, it is growing with the participation of many experts from the world, and steadily achieving its goals based on the active discussions.
With regard to what makes the essence of PIC/S, Dr Fujiwara added he thought PIC/S is ‘The core of the international harmonisation of GMP standards, and effective international cooperation’. GMP inspection authorities are responsible for the quality and stable supply of pharmaceuticals which are delivered to patients through very complicated supply chains. It is impossible for a single regulatory authority to understand complicated systems correctly, and lead to adequate and timely regulatory guidance. It is an effective ‘International harmonisation and cooperation’ among regulatory authorities that protects health and lives of the patients.
He expected participants to have active discussions in this seminar, and to enhance building ‘International harmonisation and cooperation’ while emphasizing that PMDA will continue contributing to the activities of PIC/S.
|
|
The 2.5 day Seminar consisted of a mix of presentations and parallel workshops. The first day addressed the ongoing revision of Annex 1 with a summary of the revision and points discussed. This was presented by both the PIC/S-EMA Working Group Chair (France / ANSM) and a Member (UK / MHRA), on the revision of Annex 1. The following two topics were then addressed in two presentations followed by two parallel workshops:
- Process Simulation Test (PST) (presentation by US FDA and workshop on this topic facilitated by Austria / AGES with co-facilitation by Japan / PMDA);
- Clean Room Management for Sterile Products (presentation by Canada / ROEB) followed by Clean Room Qualification and Environmental Monitoring (workshop facilitated by Switzerland / Swissmedic, with co-facilitation by Singapore / HSA).
|
|
|
|
|
|
The second day focused on Risk Management for Sterile Products (presentation by Ireland / HPRA), Quality Control and Stable Supply of Single-Use Systems (presentation by invited speaker from team member of regulatory science group) and Management of Water for Injection (WFI) (presentation by invited manufacturer association speaker) followed by two parallel workshops on:
- Single Use System (workshop facilitated by Japan / PMDA with co-facilitation by Chinese Taipei / TFDA and Japanese Prefecture of Toyama);
- Restricted Access Barriers Technology (RABS) and Isolators (workshop facilitated by Japan / PMDA with co-facilitation by Thailand / Thai FDA and Japanese Prefecture of Saitama).
The outcome and feedback resulting from these workshops were discussed during a panel discussion on the last third day, preceded by a series of presentations dedicated to Examples of observations for Sterile Products (Australia / TGA) and Rapid Microbiological Methods (invited industry association).
|
|
*****
|
| |
 |
| |
|
|