| |
|
|
|
|
Press release: PIC/S Meetings in Taipei
From 11 to 15 September 2017, the Taiwan Food and Drug Administration (TFDA) hosted the following events in Taipei (Chinese Taipei): PIC/S Committee meeting, PIC/S Executive Bureau meeting and PIC/S Annual Seminar.
|
HEADLINES
|
- NEW PIC/S CHAIRMAN, NEW EXECUTIVE BUREAU AND NEW SUB-COMMITTEE MEMBERS FOR 2018-2019
- IRAN / IFDA, TURKEY / TMMDA AND MEXICO / COFEPRIS JOIN PIC/S
- PIC/S 2017 SEMINAR ON "QUALITY CONTROL LABORATORIES: HOW TO INSPECT" HOSTED BY CHINESE TAIPEI / TFDA DRAWS MORE THAN 170 INSPECTORS FROM 50 COUNTRIES
- SAUDI ARABIA / SFDA AS WELL AS RUSSIAN FEDERATION / MINPROMTORG & FSI SID&GP APPLY FOR PRE-ACCESSION; ARMENIA / SCDMTE APPLIES FOR ACCESSION
- PIC/S VISION AND PERSPECTIVES FOR 2018-2020
- NEW TOOL AND FRAMEWORK TO STRENGHTEN RELIANCE AND MAXIMISE RESOURCES FOR GMP COMPLIANCE OF OVERSEAS FACILITIES
- CONTINUED CONCERNS WITH DRAFT EU GMP GUIDELINES FOR ATMPS
- LATEST PIC/S DEVELOPMENTS IN THE FIELD OF GM(D)P HARMONISATION
|
PIC/S COMMITTEE MEETING
|
 |
|
The PIC/S Committee met on 11-12 September 2017, under the chairmanship of Mr Paul Hargreaves (United Kingdom’s Medicines and Healthcare products Regulatory Agency / MHRA). The meeting was attended by 40 out of 49 PIC/S Participating Authorities (PA) as well as by a number of Applicants, Pre-Applicants, and Associated Partner Organisations. |
|
NEW PIC/S CHAIRMAN
|
|
The PIC/S Committee elected Mr Boon Meow Hoe (Singapore / HSA) as Chairman for the period 2018-2019. Mr Boon has been an active member of the PIC/S Committee since 2001. He has also served as a PIC/S Executive Bureau member since 2010 and is the visionary of the PIC/S Inspectorates' Academy (PIA). He will be the first PIC/S Chairman from Asia.
The incoming Chairman thanked the Committee and paid tribute to the outgoing Chairman, Mr Paul Hargreaves (UK / MHRA). Mr Hargreaves has been involved with PIC/S since 1986 and his chairmanship was marked by an innovative approach, outlining the future of PIC/S and contributing to a significant raise of profile of PIC/S at an international level. PIC/S membership has successfully increased during his term (2016-2017).
|
|
 Outgoing PIC/S Chairman (2016-2017), Outgoing PIC/S Chairman (2016-2017),
Mr Paul Hargreaves (UK / MHRA), welcoming incoming PIC/S Chairman (2018-2019), Mr Boon Meow Hoe (Singapore / HSA). |
The PIC/S Committee elected as incoming Deputy Chairperson for the period 2018-2019, Ms Anne Hayes (Ireland / HPRA), who will continue to chair the Sub-Committee on Compliance (SCC), one of the largest PIC/S Sub-Committees, in charge of compliance with the PIC Scheme.
The incoming Chairman paid also tribute to another long-standing member of the Committee, former PIC/S Chairman (2006-2009) and current Chair of the Sub-Committee on Strategic Development (2014-2017), Mr Jacques Morénas (France / ANSM), who will be retiring from his Agency in the course of 2018. Mr Morénas actively contributed to PIC/S over the years, in particular to PIC/S expansion through his involvement in a great many of PIC/S accession assessments.
|
NEW PIC/S SUB-COMMITTEE OFFICE HOLDERS
|
|
The PIC/S Committee elected the Members, Deputy Chairs and Chairs of the PIC/S Sub-Committee structure for the period 2018-2019. Office holders were elected for the following seven Sub-Committees: Training (SCT); Expert Circles (SCEC); Strategic Development (SCSD); Compliance (SCC); GM(D)P Harmonisation (SCH); Budget, Risk and Audit (SCB) and Communication (SC COM). All Sub-Committee Chairs will be Members of the PIC/S Executive Bureau. Out of the 75 newly appointed Sub-Committee office holders, all continents are represented and 39 are from non-EEA PIC/S Participating Authorities.
This represents a significant increase in the number of office holders - up from 52 in 2015. The increase reflects the growing scope of PIC/S activities, in particular its working groups. It also speaks to the success encountered by PIC/S as a more participative and efficient organisation as implemented in 2014 with the new Sub-Committee structure. Relevant experience, competence and suitability, as well as the need for a balanced regional representation and a balanced participation amongst Participating Authorities, are among the criteria taken into account for these elections.
|
NEW PIC/S EXECUTIVE BUREAU
|
|
A new PIC/S Executive Bureau was elected by the Committee as from 1 January 2018. The Executive Bureau Members for the period 2018- 2019 are:
• Mr Boon Meow Hoe (Singapore / HSA), PIC/S Chairman;
• Ms Anne Hayes (Ireland / HPRA), PIC/S Deputy Chairperson and Chair of the Sub-Committee on Compliance (SCC);
• Mr Paul Hargreaves (United Kingdom / MHRA), Immediate Past PIC/S Chairman;
• Mr Jacques Morénas (France / ANSM), Chair of
the Sub-Committee on Training (SCT), until his retirement;
• Mr Paul Gustafson (Health Canada), Chair of the
Sub-Committee on GM(D)P Harmonisation (SCH);
|
• Dr Andreas Krassnigg (Austria / AGES), Chair of the Sub-Committee on Expert Circles (SCEC);
• Mr Ger Jan van Ringen (Netherlands / IGZ), Chair of the Sub-Committee on Budget, Risk and Audit (SCB);
• Mr Mark Birse (United Kingdom / MHRA), Chair of the Sub-Committee on Communication (SC COM);
• Ms Susan Laska (US FDA), Chair of the Sub-Committee on Strategic Development (SCSD).
|
|
IRAN / IFDA
TURKEY / TMMDA
AND MEXICO / COFEPRIS
JOIN PIC/S
|
|
The PIC/S Committee invited 3 new Authorities to join the Scheme as from 1 January 2018, namely the Iran Food and Drug Administration (IFDA), the Turkish Medicines and Medical Devices Agency (TMMDA), and Mexico's Federal Commission for the Protection from Sanitary Risks (COFEPRIS).
|
|
Iran / IFDA will become PIC/S’ 50th Participating Authority. IFDA (previously Iran / MoH) submitted a complete membership application in September 2010. A paper assessment was conducted in 2012 in view of its accession to PIC/S. Due to changes in Rapporteur and other changes affecting the Applicant, which led to a 1 year clock-stop, the on-site visit took place in September 2015, followed by a follow-up visit in July 2017.
The follow-up visit allowed to review the implementation of CAPA and successfully finalise the assessment. The Audit team recommended to the Committee to accept the PIC/S membership application of Iran / IFDA.
The representatives of Iran / IFDA, Dr Amin Motallebyghomi (3) and Dr Zohre Bazaz (4), with the PIC/S Chairman, Mr Paul Hargreaves (UK / MHRA) (5) and the PIC/S Deputy Chairman, Mr Boon Meow Hoe (Singapore / HSA) (1) and PIC/S Chair of the Sub-Committee on Compliance, Ms Anne Hayes (Ireland / HPRA) (2).
|
Turkey / TMMDA will become PIC/S’ 51st Participating Authority. TMMDA applied for PIC/S membership in May 2013. A paper assessment was conducted over the period 2014-2015 in view of its accession to PIC/S. The on-site visit took place in February 2017.
The Audit team recommended to the Committee to accept the PIC/S membership application of Turkey / TMMDA.
| The representatives of Turkey / TMMDA, Mr Fatih Tan (5), Ms Gülsen Yilmaz (3), Mr Muhammed Enes Demir (6), with the PIC/S Chairman, Mr Paul Hargreaves (UK/MHRA) (4), the PIC/S Deputy Chairman, Mr Boon Meow Hoe (Singapore / HSA) (2) and the Rapporteur as well as PIC/S Chair of the Sub-Committee on Compliance, Ms Anne Hayes (Ireland / HPRA) (1). |

|
|
|
Mexico / COFEPRIS will become PIC/S’ 52nd Participating Authority. COFEPRIS submitted a PIC/S pre-accession application in May 2013. Further to the completion of the gap-analysis, COFEPRIS applied for PIC/S membership in December 2014.
An on-site visit took place in January 2016, followed by a follow-up visit in July 2017 in order to assess implementation of the CAPA. The accession of COFEPRIS was considerably facilitated through the pre-accession process undergone previously. COFEPRIS is the first new Member to successfully join PIC/S after the introduction of the pre-accession procedure. The Audit team recommended to the Committee to accept the PIC/S membership application of Mexico / COFEPRIS.

|
The representatives of Mexico / COFEPRIS, Mr Mario Alanís (5); Mr Marcos Solis (2) and Mr Mario Perez (8); with the PIC/S Chairman, Mr Paul Hargreaves (UK / MHRA) (4); the PIC/S Deputy Chairman, Mr Boon Meow Hoe (Singapore / HSA) (1); the PIC/S Chair of the Sub-Committee on Compliance, Ms Anne Hayes (Ireland / HPRA) (3); the Rapporteur, Mr Manuel Ibarra (Spain / AEMPS) (6); and one of the assessment team members, Ms Marisa Delbò (Italy / AIFA) (7).
|
|
2017 PIC/S ANNUAL SEMINAR ON
QUALITY CONTROL LABORATORIES: HOW TO INSPECT
|
 Deputy Minister, Ministry of Health and Welfare, Deputy Minister, Ministry of Health and Welfare,
Dr Chi-Kung Ho (left) with Director-General of TFDA, Dr Shou-Mei Wu (right) |
The 2017 PIC/S Annual Seminar was organised by Taiwan Food and Drug Administration, Ministry of Health and Welfare (TFDA) in Taipei (Chinese Taipei) on 13-15 September 2017. The topic of the Seminar was “Quality Control Laboratories: How to Inspect”. The Seminar was opened by the Deputy Minister, Ministry of Health and Welfare, Dr Chi-Kung Ho, along with the Director-General of TFDA, Dr Shou-Mei Wu, and the PIC/S Chairman, Mr Paul Hargreaves.
|
The Seminar, which was the first organised in Chinese Taipei since TFDA joined PIC/S in 2013, was attended by more than 170 inspectors from more than 50 countries. All continents were represented.
Quality Control is part of Good Manufacturing Practice and ensures the safety, quality and efficacy of pharmaceutical products. Consequently, Quality Control Laboratories (QC labs) play an integral part in confirming the quality of medicinal products. Through this Seminar, which consisted in a mix of presentations and workshops, participants were introduced to basic concepts, current and trending regulatory requirements, as well as specific quality issues which have an impact on regulatory compliance of QC labs.
The Seminar programme covered topics related to GMP inspections of QC labs and delved into Out-of-Specification/Out-of-Trend investigations, data integrity issues and aspects of test methods validation and verification in the context of technical transfers.
The Seminar also offered a great platform for both novice and experienced inspectors to enhance their inspection skills through knowledge sharing and discussion. In particular, it facilitated discussions and the sharing of inspection experiences, with a focus on frequent non-compliances and special cases observed during inspection of laboratories.
It also allowed inspectors to advance their inspection skills and the implementation of risk assessments and inspection strategies during the inspection of chemical and microbiological laboratories. Finally, it aimed at revising the current PIC/S Aide Memoire on inspection of pharmaceutical quality control laboratories (PI 023-2), which entered into force on 1 January 2006.
For more information on the Seminar, see below.
|
SAUDI ARABIA / SFDA AND
RUSSIAN FEDERATION / MINPROMTORG AND FSI SID&GP
APPLY FOR PRE-ACCESSION
ARMENIA / SCDMTE APPLIES FOR ACCESSION
|
|
The Saudi Food and Drug Authority (SFDA) applied for PIC/S pre-accession on 31 July 2017. A Rapporteur and Co-Rapporteur were appointed by the Committee.
The Ministry of Industry and Trade of the Russian Federation (Minpromtorg Russia) jointly with the Federal State Institution “State Institute of Drugs and Good Practices” (FSI “SID & GP”) applied for PIC/S pre-accession on 28 August 2017. A meeting took place on 11 September 2017 in Taipei between the PIC/S Executive Bureau and a Russian Delegation. Discussions provided an opportunity for better understanding of the Russian organisation of GMP inspectorates as well as of PIC/S requirements, respectively. A Rapporteur and Co-Rapporteur were also appointed by the Committee.
Armenia's Scientific Centre of Drug and Medical Technology Expertise (SCDMTE) applied for PIC/S accession on 8 September 2017, further to addressing the CAPA resulting from the outcome of the pre-accession procedure.
|
PIC/S VISION AND PERSPECTIVES FOR 2018-2020
|
|
The Committee reviewed and adopted a PIC/S Road-Map which is a non-binding policy paper highlighting the possible future strategic orientations of PIC/S over the next three years (2018-2020).
In line with PIC/S mission which is “to lead the international development, implementation and maintenance of harmonised GMP standards and quality systems of inspectorates in the field of medicinal products”, the Road-Map will guide the work of the Organisation and its Members and allow them to further develop the following goals:
- Offering a training academy to all inspectors by means of the PIC/S Inspectorates’ Academy (PIA);
- Facilitating the exchange of GMP information by mutual confidence based on the equivalence of PIC/S Participating Authorities (i.e. Members);
- Identifying PIC/S’ next challenges and addressing them, notably by:
|
- Enhancing PIC/S’ Sub-Committee (SC) structure;
- Strengthening the PIC/S Secretariat and implementing an effective human resourcing strategy; and
- Identifying new income streams, which will yield the required funding necessary to finance PIC/S’ projects.
|
NEW TOOL AND FRAMEWORK TO STRENGHTEN RELIANCE
AND MAXIMISE RESOURCES FOR GMP COMPLIANCE
OF OVERSEAS FACILITIES
|
|
Among its future orientations, the Committee agreed to take over the GMP project of the International Coalition of Medicines Regulatory Authorities (ICMRA) upon request of the latter. This GMP project consists in a new tool (standard operating procedure) developed and tested by the ICMRA. The aim of this procedure, applicable on a voluntary basis, is to create a framework to maximise inspection resources to enable increased reliance on other trusted Regulatory Authorities for the GMP compliance of overseas facilities.
The Committee decided to carry out a consultation of PIC/S Participating Authorities in order to review and adapt this tool to PIC/S' needs. Further to this consultation, a PIC/S Working Group may be established in order to further refine the tool, in view of its future adoption and implementation by PIC/S, which has the support of ICMRA Heads of Agencies.
|
CONTINUED CONCERNS WITH
DRAFT EU GMP GUIDELINES FOR ATMPS
|
|
The Committee discussed the outcome of a teleconference meeting of 4 July 2017 between the European Commission (EC) and the PIC/S ad-hoc Advanced Therapies Medicinal Products (ATMPs) drafting group set up to respond to the 2016 EU targeted stakeholder consultation of EU draft Guidelines on GMP for ATMPs. Further to the strong stance taken by PIC/S on this initiative by the EU (see PIC/S' press release of March 2017), the Committee noted that while progress has been made by the EC in addressing some of the issues raised by PIC/S, a number of serious patient safety concerns remain.
Some PIC/S Participating Authorities will also continue to raise remaining concerns directly with the EC. It is hoped that the EC will be responsive to these concerns in order to ensure equivalent standards to those of PIC/S. A strategy for possible harmonisation with EU GMP ATMP requirements will need to be discussed by PIC/S once the new requirements will have been published by the EC.
|
LATEST PIC/S DEVELOPMENTS IN THE FIELD
OF GM(D)P HARMONISATION
|
| The revision of Chapter 3, 5, and 8 of the PIC/S GMP Guide are ongoing as well as the transposition by PIC/S of the EMA Guidelines on: |
| |
- the formalised risk assessment for ascertaining the appropriate good manufacturing practice for excipients of medicinal products for human use;
- the principles of good distribution practice for active substances of medicinal products for human use;
- setting health based exposure limits for use in risk identification in the manufacture of different medicinal products in shared facilities.
|
|
Feedback is awaited from the European Commission on the draft revision of Annex 1 (sterile manufacturing) by the PIC/S – EMA Working Group on Annex 1, chaired by UK / MHRA, in order for the EMA to launch jointly with PIC/S and WHO the public consultation.
An update on the status of the work of the EMA drafting groups in which PIC/S is represented for the revision of Annex 13 (Investigational Medicinal Products), Annex 17 (Real time release testing) and for (new) Annex 21 (Imports) was provided as well as on progress in the revision of procedures for quality defects and Rapid Alerts by a joint PIC/S - EMA Drafting Group.
Further to the proposal to have a PIC/S version of EU Annex 16 (Certification by a QP & Batch Release) a PIC/S Drafting Group will be established in the near future.
A Drafting Group, led by Italy / AIFA, was established by the Committee for the revision of the PIC/S GMP Guide for Blood Establishments (PE 005-3) and PIC/S Guide to Inspections of Source Plasma Establishment and Plasma Warehouses (PE 008-3), with the aim to merge and harmonise these with the EU Good Practices Guidelines for blood establishments.
The Committee was updated on recent work by the Working Group on Data Integrity, co-chaired by UK / MHRA and Australia / TGA, in particular the development of a revised draft of the PIC/S guidance on Good Practices for Data Management and Integrity in Regulated GMP/GDP Environments, for which a public consultation and a new trial-implementation period will be organised at a later stage. Other data integrity guidance tools for inspectors are currently under development.
A revision of the PIC/S Recommendations on Validation Master Plan; Installation and Operational Qualification; Non-Sterile Process Validation; and Cleaning Validation (PI 006-3) is underway.
A revised draft of the PIC/S Guidance on Classification of Deficiencies has been developed by the Working Group on Harmonisation of Classification of Deficiencies, chaired by Australia / TGA. This revised draft was the focus of one of the 2017 PIC/S Seminar workshops (see below).
The adoption of a PIC/S Aide Memoire on Cross Contamination in Shared Facilities, developed by the Working Group on Controlling Cross Contamination in Shared Facilities, chaired by UK / MHRA, and of a PIC/S Aide-Memoire on Inspection of Manufacturers and Wholesale Distributors for Compliance with GDP and draft Q&A for the PIC/S GDP Guide, developed by the Expert Circle on GDP, also chaired by UK / MHRA, will be carried out by written procedure.
|
|
PIC/S MEMBERSHIP APPLICATIONS
|
|
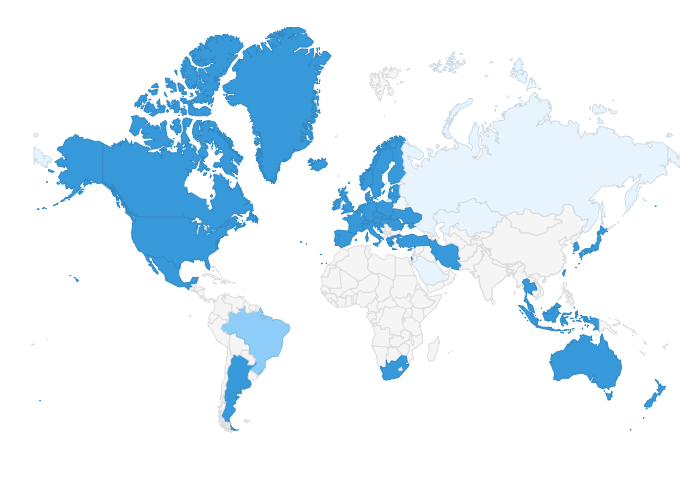
|
| Overview of PIC/S 52 Members as of 1 January 2018 (dark blue) including Iran / IFDA, Turkey / TMMDA and Mexico / COFEPRIS; 2 Applicants (medium blue) and 3 (Pre-) Applicants (pale blue) |
| |
Applicants (2) |
Pre-Applicants (3) |
| |
Brazil / ANVISA
Italy (Vet) / DGSAF
Armenia / SCDMTE*
|
Kazakhstan / CCMPA
Russian Federation / Minpromtorg and
FSI SID&GP
Saudi Arabia / SFDA
|
|
|
- A call for Rapporteurs was made for the assessment of the membership application of Armenia / SCDMTE, once it is completed;
- Brazil / ANVISA will be re-lodging its membership application further to a major re-organisation;
- The Committee was updated on the status of the membership application of Italy (Vet) / DGSAF, which will be facilitated by a partial assessment, taking into account the audit performed within the EMA Joint Audit Programme, in line with the PIC/S-EEA HMA Letter of Agreement;
- The Committee reviewed the pre-accession gap-analysis report for Kazakhstan / CCMPA, further to which CCMPA will have the opportunity to provide a CAPA.
|
PIC/S INSPECTORATES' ACADEMY
|
|
The PIC/S Inspectorates' Academy (PIA) is an initiative for a web-based educational centre under the PIC/S umbrella, which aims at harmonising and standardising GMP training at an international level through a certified qualification system. The PIA website was launched in July 2016 and aims to deliver not only general or advanced training but also serve as a platform for discussion and sharing among regulators. It offers a single point of access to all PIC/S training activities and is being implemented in various stages.
Ireland / HPRA reported on the prospect of institutionalising and regularly running the PIC/S New Inspector Training Course. This course is highly appreciated by new inspectors and has been identified as key training in the frame of the PIA.
The Committee was also updated on the development of new PIC/S video training materials, on the trial of new training tools such as webinars, on projects for future on-line training and on opportunities for co-operation with Professional Associations (e.g. ISPE and PDA). A teleconference meeting with ISPE took place on 30 May 2017.
|
OTHER NEWS
|
The PIC/S Committee:
|
- adopted the mandate of the new PIC/S Working Group on Unique Facility Identifiers (UFI), chaired by US FDA;
- adopted a new mandate for the PIC/S Expert Circle on Quality Risk Management (QRM), chaired by Austria / AGES;
- adopted new Guidelines for the PIC/S Joint Visits Programme (JVP) in the field of Good Clinical Practices (GCP) & Good Pharmacovigilance (GVP), developed by the PIC/S Working Group on GCP & GVP, chaired by UK / MHRA. The JVP allows 3 Inspectors from 3 different countries to team up to observe inspections in each country with a view to comparing inspection procedures and techniques and harmonising interpretation in their field of competence;
- discussed the topic of the 2018 PIC/S Seminar on "Assessment of Risk through the Product Life-Cycle" which will be hosted by US FDA in Chicago (USA) on 26-28 September 2018;
 |
Passing of the PIC/S walking stick, which symbolises the hosting of a PIC/S Seminar, from the 2017 Seminar host TFDA, represented by the Director-General of TFDA, Dr Shou-Mei Wu to the 2018 Seminar host US FDA, represented by US FDA Assistant Commissioner for Medical Products and Tobacco Operations, Ms Ellen F. Morrison, surrounded by PIC/S Chairman, Mr Paul Hargreaves (UK/MHRA) (left) and PIC/S Deputy Chairman, Mr Boon Meow Hoe (Singapore/HSA) (right). |
- accepted an invitation from Japan / PMDA & MHLW to host the 2019 PIC/S Seminar on the foreseen topic of "Quality Assurance of Sterile Medicinal Products - Annex 1" in Japan in October 2019;
- discussed the planning of the PIC/S re-assessments of Argentina / INAME, Switzerland / Swissmedic and Ukraine / SMDC scheduled for 2018 and appointed the PIC/S Re-Assessment Team members;
- noted the PIC/S re-assessments of Australia / TGA and Singapore / HSA which will have taken place on 18-22 September 2017;
- was updated on the status of the guideline and interpretation of the Audit Checklist by the PIC/S Sub-Committee on Compliance, in co-operation with the EU Joint Audit Programme Compliance Group;
- was updated by the PIC/S – ASEAN Liaison Authority, Thailand / Thai FDA, on recent activities within ASEAN. The Committee was also informed that the ASEAN Pharmaceutical Product Working Group (PPWG) has considered positively the proposal to establish a light instrument for co-operation between PIC/S and the ASEAN PPWG in specific areas of mutual interest;
- was updated on current developments with regard to the implementation of the EU – US Mutual Recognition Agreement (MRA) and was updated on recent GMP-related developments by EDQM, EMA and UNICEF as PIC/S Associated Partner Organisations;
- was updated on the development of a PIC/S' stakeholders' mapping tool;
- noted the entry into force of the revised PIC/S Rapid Alert Procedure (PI 010-5) on 1 July 2017, which was revised in order to maintain its alignment with the current EMA rapid alert procedure;
- noted that ICH approved PIC/S as an ICH Observer;
- noted the status of the new Working Group on Inspector Travel Safety, led by Australia / TGA; the status of the PIC/S Working Group (WG) on Veterinary Medicinal Products, led by France / ANSES and UK / VMD and the status of the PIC/S Working Group on the API Q&A established for the development of training material part of the PIC/S API International Training Programme based on API Q&A which were not integrated into the ICH Q7 Q&A document, led by Switzerland / Swissmedic.
|
RECENT TRAINING ACTIVITIES
|
|
The 8th PIC/S Expert Circle on APIs meeting and Advanced Training was held in Melbourne, on 5-7 April 2017, hosted by Australia / TGA. The meeting was dedicated to strengthening international co-operation, advancing knowledge and sharing experiences in the field of API manufacturing processes and inspections. The main topic of the meeting was regulation of small versus large molecule manufacturing. Other topics discussed included regulatory approaches to atypical API’s, continuous processing and EU guidance on good distribution practices. The programme included 2 workshops on atypical APIs and on the review of the PIC/S Aide Memoire on the inspection of APIs (PI 030-1). The training also provided the opportunity of a visit of a biotech manufacturing facility. The meeting was attended by more than 70 participants from 24 authorities and international organisations.

|

The 23rd PIC/S Expert Circle on Human Blood, Tissues, Cells and ATMPs was held in Seoul, on 26-28 June 2017, hosted by Korea (Republic of) / MFDS. The meeting provided an opportunity for expert discussions on contemporary issues in the field of blood, plasma derivatives, cells and tissues, with particular focus on ATMPs. The meeting allowed for an update on the mapping of competences of PIC/S Participating Authorities in these fields as well as on the latest regulatory developments concerning the draft EU GMPs for ATMPs. The meeting offered also training in the fields of blood derivatives, tissues and ATMPs through visiting manufacturing facilities in Korea and facilitated sharing of experiences between inspectors with a view to improve consistency. The meeting was attended by more than 70 participants from 25 authorities. |
|
|
A GMP Training Seminar organised by PMDA Asia Training Center for Pharmaceuticals and Medical Devices Regulatory Affairs (PMDA-ATC) was held on 31 July - 4 August 2017 in Yamaguchi, Japan. This training event was supported by PIC/S.
|
COMING UP...
|
|

|
- 23-27 October 2017: PIC/S New Inspector Training Course, in Dublin (Ireland), hosted by Ireland / HPRA;
- 26-28 September 2018: Annual PIC/S Seminar on “Assessment of Risk through the Product Life-Cycle”, in Chicago (USA), hosted by US FDA;
- PIC/S Expert Circle on Blood, Tissues, Cells and ATMPs, in Warsaw (Poland), hosted by Poland / CPI (dates to be confirmed);
- PIC/S Expert Circle on Good Distribution Practices, hosted by Spain / AEMPS (dates and location to be confirmed);
- 2018 Japan / PMDA - ATC GMP Inspection Seminar, with the support of PIC/S, in Japan, hosted by PMDA (dates to be confirmed).
|
|
|
2017 PIC/S ANNUAL SEMINAR ON QUALITY CONTROL LABORATORIES:
HOW TO INSPECT
|
|

Deputy Minister, Ministry of Health and Welfare, Dr Chi-Kung Ho
|
Seminar Opening Address by
Deputy Minister, Ministry of Health and Welfare,
Dr Chi-Kung Ho
Dr Chi-Kung Ho welcomed all Seminar participants to Taipei. He gave an overview of the history and achievements accomplished over the years in upgrading the GMP system in line with international standards, highlighting some milestones such as the adoption of the PIC/S GMP Guide. It was an honour for the Taiwan Food and Drug Administration (TFDA), which is under the Ministry of Health and Welfare since 2013, to host this year's Annual Seminar. He highlighted the efforts which went into the organisation of this Seminar, its topics, workshops and expected outcome. He welcomed this opportunity for international co-operation between regulators.
|
|
|
The 2.5 day Seminar consisted of a mix of presentations and parallel workshops. The first day started with a series of lectures and presentations on current GMP requirements for the inspection of Quality Control Laboratories, in particular for physical, microbiological and analytical laboratories, followed by two parallel workshops on:
- the PIC/S Aide Memoire on Inspection of Quality Control Laboratories (PI 023-2) which allowed for discussion and input in view of a future revision of this guidance document (Workshop leaders: Health Canada and Chinese Taipei / TFDA);
- the Harmonisation of the Classification of Deficiencies of Quality Control, which included practicising the application of the risk based management tool part of the guidance currently in development by the PIC/S Working Group on Harmonisation of Classification of Deficiencies (Workshop leaders: Australia / TGA and Croatia / HALMED).
The second day delved into specific quality control issues, with presentations on Out-of- Specification (OOS) and Out-of-Trend (OOT) Investigations - expectations and commonly found deficiencies as well as on Data Integrity in Quality Control, from the perspective of an Inspector and from that of an invited Industry guest, followed by two parallel workshops on:
- the discussion and review of a Data Integrity case study (Workshop leaders: US FDA and Japan / PMDA);
- the discussion and review of an Out-of- Specification / Out-of-Trend case study (Workshop leader: UK / MHRA).
The outcome and feedback resulting from these workshops were discussed on the last day, followed by a series of presentations on Lifecycle Management of Analytical Procedures including Test Methods Validation and Methods Verification in the context of a Technical Transfer. The perspectives from an Inspector, an invited guest Reviewer and from an invited guest Industry speaker were given at this occasion.
|
| ***** |
 |
| |
|
|